
Because of the strong electrostatic interaction between these oppositely charged ions, ionic compounds form rigid crystals. A certain number of anions encircle each cation and vice versa. Why do ionic compounds form hard crystals?Ĭrystal lattices having oppositely charged ions make up the crystals of electrovalent substances. 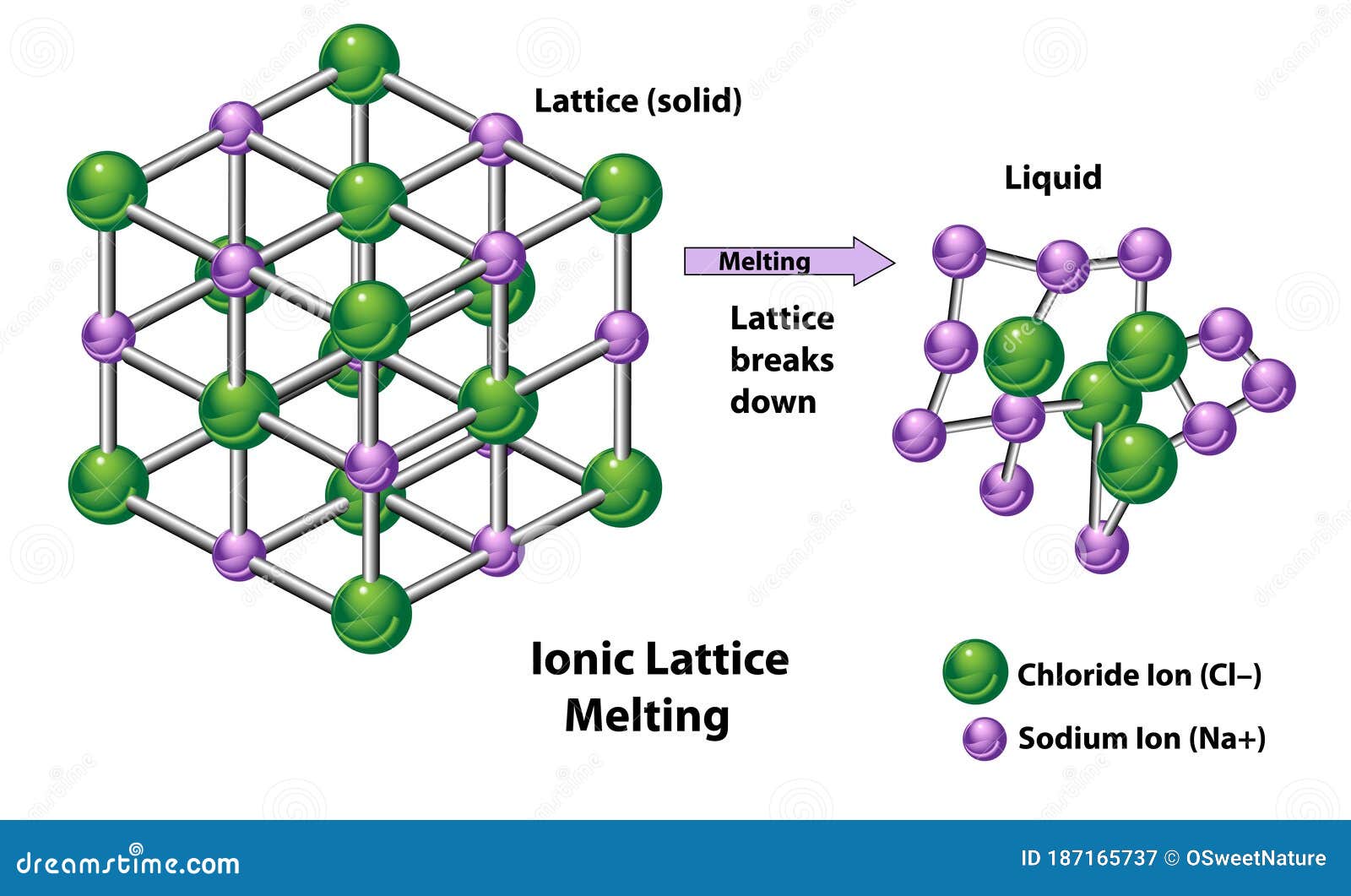
Many consecutive positive and negative ions are linked in a matrix to form a crystal.Ģ. Ionic chemicals form crystals rather than molecules. Crystals of ionic compounds have high melting and boiling points.Metals transmit valence electrons to non-metals to generate ionic compounds.Ionic compounds have a total net neutral charge because the opposite charges balance each other out.Ionic compounds are made up of ions with opposing charges kept together by ionic bonds.Other common uses of ionic compounds are shown below: While a salt crystal is an electrical insulator, saline fluids (salt mixed in water) do not. Table salt, often known as sodium chloride, is an ionic substance, and the melting point of salt is very high. The dispersed ions in ionic compounds mixed with water can carry electric current through the mixture.When mixed with water, they conduct electricity.Ionic crystals are difficult to distinguish because the positive and negative ions are firmly attached.High temperatures are demanded to counteract the affinity between oppositely charged ions in ionic compounds. They have very high melting and boiling points.An ionic crystal is a regular pattern at the atomic scale, with the cation and anion cycling and generating a three-dimensional framework based on the smaller ion uniformly filling up the spaces between the bigger ions. Though molecular substances may form crystals, they generally do so in different shapes than ionic compounds, and molecular crystals are generally smoother than crystalline materials. Instead of amorphous solids, ionic compounds produce crystal lattices. Metals and non-metals commonly form ionic connections. Cations tend to get a positive charge, whereas anions have a negative charge. Nonmetals acquire negative charge by losing electrons and forming anions, whereas metals gain positive charge by losing electrons and forming cations. Ions are molecules that lose or gain electrons, and they will also have an opposite charge. Ionic compounds are compounds made up entirely of ions. Let’s learn about these compounds and crystals in detail. Others believe the “magic” lies in the crystal’s structural form, in which the cations and anions stack up in a predictable pattern □ These crystals are made up of ionic compounds. Crystals are thought to have magical properties by some individuals □. The ruby crystal seen here is incredibly precious, both for its aesthetic value and application in lasers. Crystals can be discovered in chemical deposits all over the world.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
